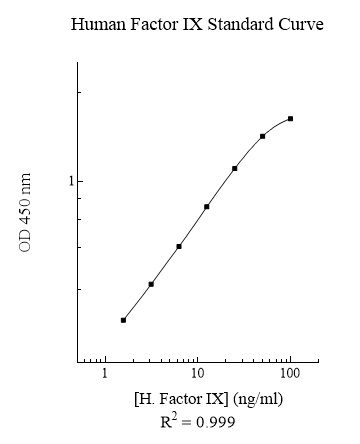
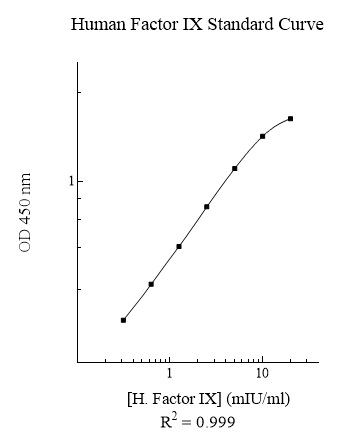
Publications (15 results)
Please use discretion when following external links; Assaypro cannot guarantee the validity and security of all citation links provided.A comprehensive model for assessment of liver stage therapies targeting Plasmodium vivax and Plasmodium falciparum
- https://www.nature.com/articles/s41467-018-04221-9
- A Roth, SP Maher, AJ Conway, R Ubalee… - Nature …, 2018 - nature.com
- Malaria liver stages represent an ideal therapeutic target with a bottleneck in parasite load and reduced clinical symptoms; however, current in vitro pre-erythrocytic (PE) models for …
* Than PA et al. (2022) Selective Microvascular Tissue Transfection Using Minicircle DNA for Systemic Delivery of Human Coagulation Factor IX in a Rat Model Using a Therapeutic Flap. Plast Reconstr Surg. 149(1):117-129
* Kruse RL (2021) Endoscopic-mediated, biliary hydrodynamic injection mediating clinically relevant levels of gene delivery in pig liver. Gastrointest Endosc. S0016-5107(21)01463-2.
* Engelmann B (2018) Plasma Proteome Analyses of Experimental Human and Murine Thyroid Disease Models. Ernst Moritz Arndt University of Greifswald. Dessertation.
* Ramaswamya S et al. (2017) Systemic delivery of factor IX messenger RNA for protein replacement therapy. Proc Natl Acad Sci USA. 114(10):E1941-E1950.
* DeRosa F et al. (2016) Therapeutic efficacy in a hemophilia B model using a biosynthetic mRNA liver depot system. Gene Therapy 23:699-
* Heartlein M et al. (2015) Lipid formulations for delivery of messenger rna. Patents: US 20150140070 A1.
* Maher SP (2014) Development of an efficient human hepatocyte culture platform for assessing novel therapeutic efficacy against Plasmodium liver parasites. University of South Florida. Graduate Theses and Dissertations.
* Heartlein M (2014) Mrna therapeutic compositions and use to treat diseases and disorders. Patent application:WO2014152940 A1
* Derosa F et al.(2014) Lipidic nanoparticles for mrna delivering. Patent application:WO2014089486 A1
* Ostroff RM et al. (2012) Early detection of malignant pleural mesothelioma in asbestos-exposed individuals with a noninvasive proteomics-based surveillance tool. PLoS One. 7(10):e46091
* Guild BC et al. (2012) Lipid nanoparticle compositions and methods for mrna delivery. Patent Application WO2012170930 A1
* Fima UE et al. (2010) Long-Acting Coagulation Factors and Methods of Producing Same. Patent No: US 8476234 B2
* Dadehbeigi N et al. (2008) Sex hormones affect the production of recombinant Factor IX in CHO and HEK-293 cell lines. Biotechnol Lett. 30(11):1909-12